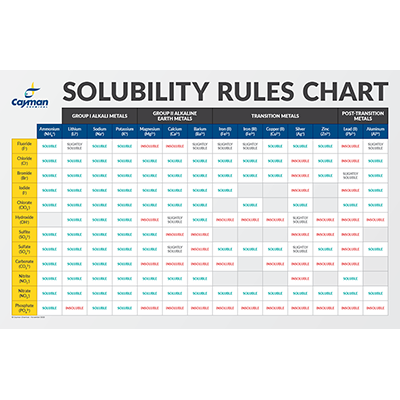
Q 1 ethanal is more soluble in water than ethane but less than the ethanol Q 2 acetone is completely miscible in water while acetophenone does not
![College Level: Inorganic Chemistry] Intermolecular Forces: Determining the solubility of Acetone/Urea in both Ethylene Glycol and O-Dichlorobenzene : r/chemhelp College Level: Inorganic Chemistry] Intermolecular Forces: Determining the solubility of Acetone/Urea in both Ethylene Glycol and O-Dichlorobenzene : r/chemhelp](https://preview.redd.it/hkmehno299e41.jpg?auto=webp&s=244b48eaddcff79edec67f1147ec068a6f32b2b8)
College Level: Inorganic Chemistry] Intermolecular Forces: Determining the solubility of Acetone/Urea in both Ethylene Glycol and O-Dichlorobenzene : r/chemhelp

SOLVED: Part V: Solubility; Like dissolves In Like: Search online for the solubility of the above solvents in water. List all solvents that are miscible [ in water from Part IV: Emanol,Ubfr ,
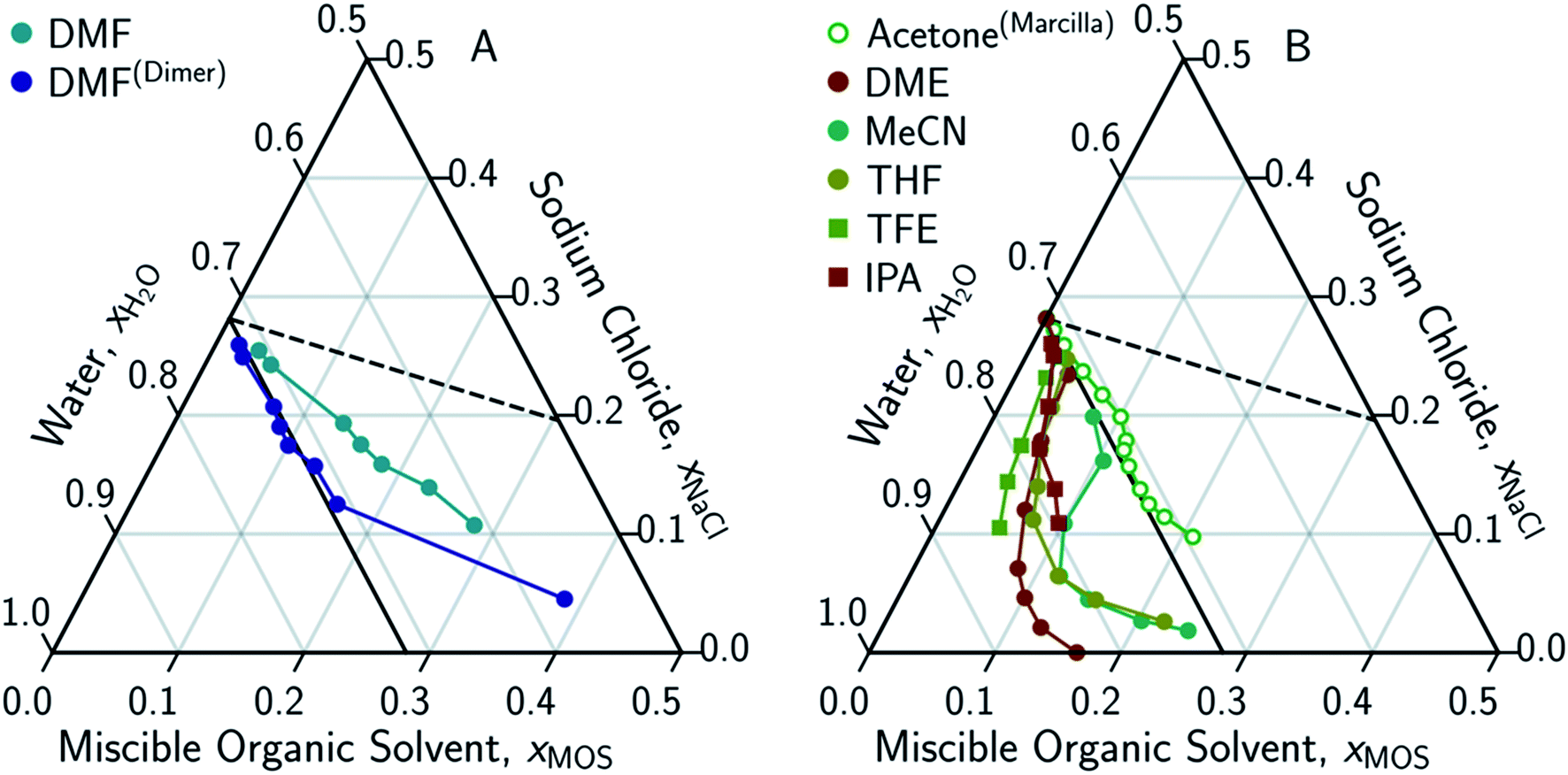
Solute displacement in the aqueous phase of water–NaCl–organic ternary mixtures relevant to solvent-driven water treatment - RSC Advances (RSC Publishing) DOI:10.1039/D0RA06361D

SOLVED: Part A Solubility Indicate if following mixtures are miscible or immiscible. cyclohexane cyclohexene toluene water acetone Which compound is less dense waler or toluene? Explain yOuI answer.

Pharmaffiliates - #Acetone is produced and disposed of in the #human_body through normal #metabolic_processes. It is miscible with water and serves as an important solvent in its own right, typically for cleaning