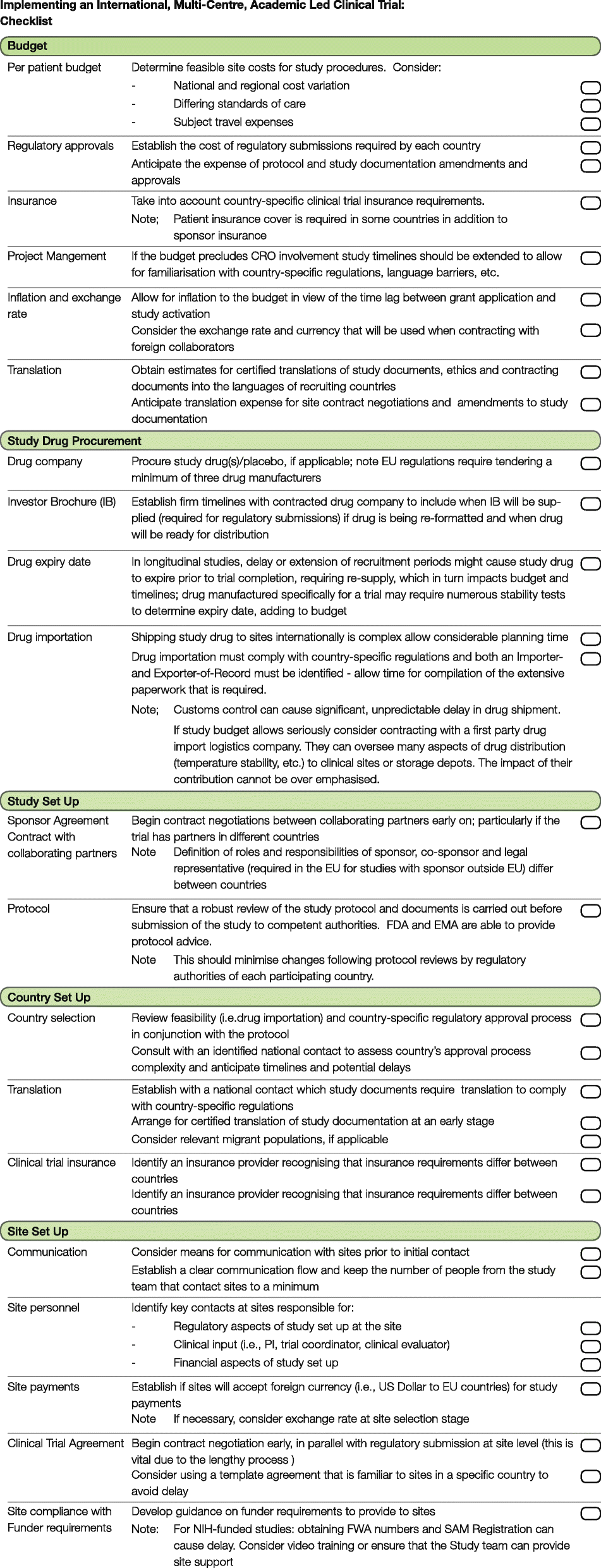
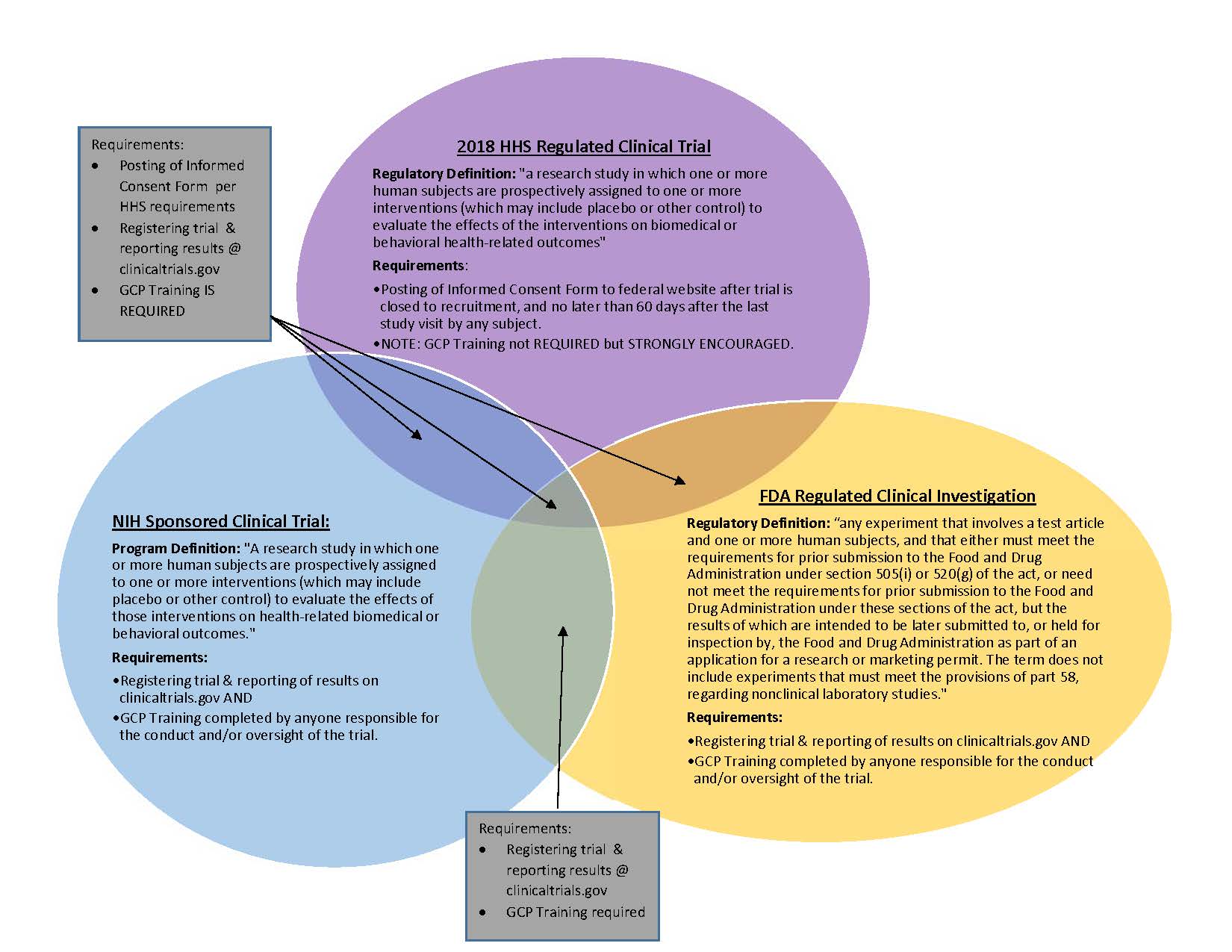
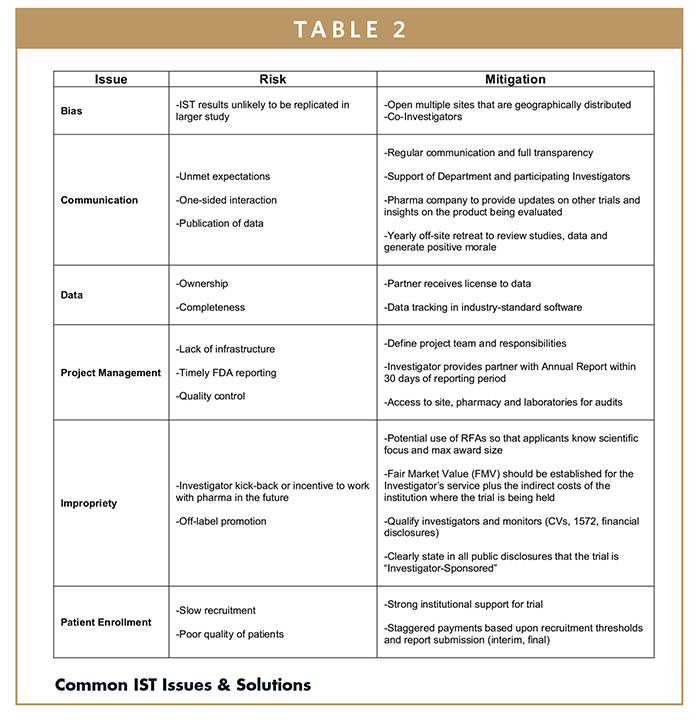
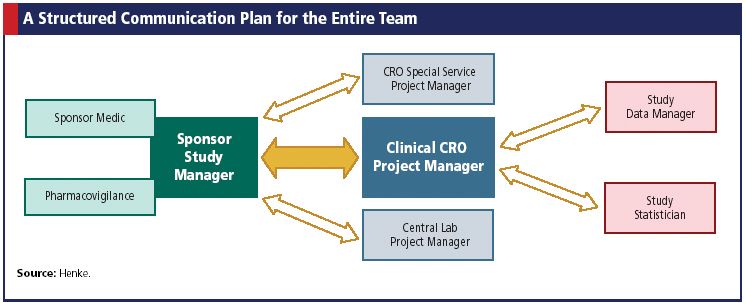
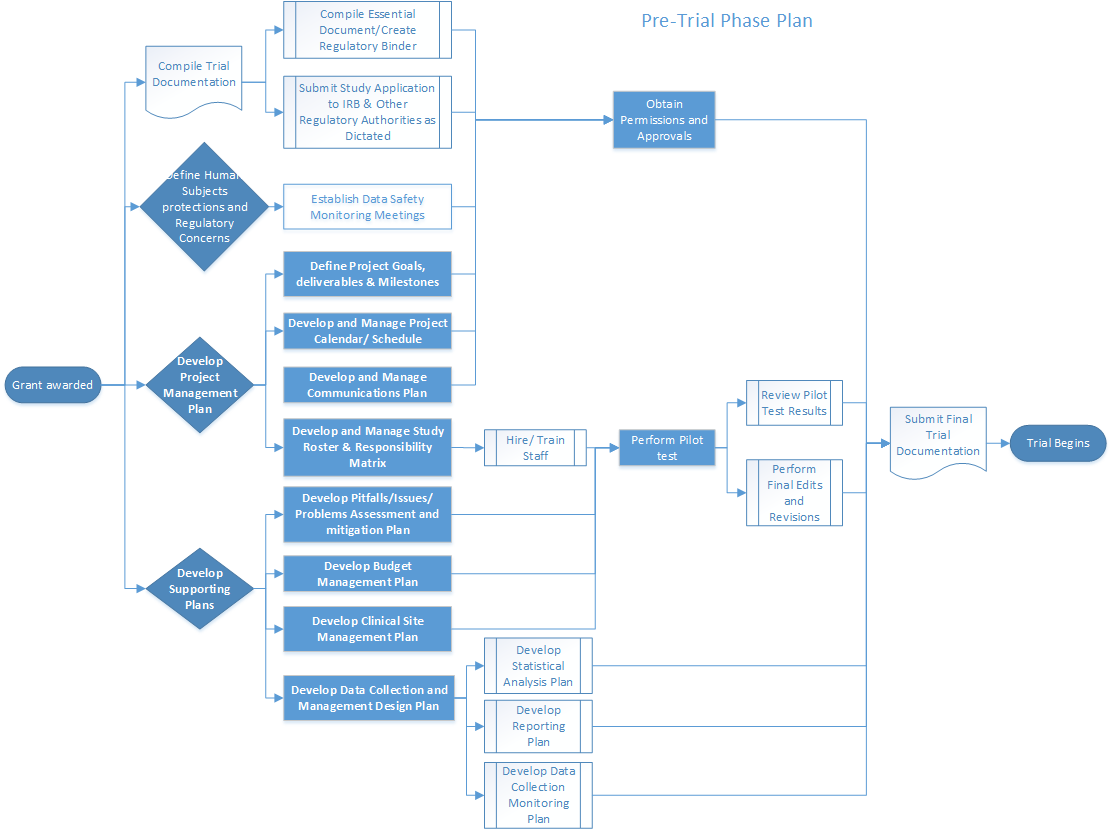
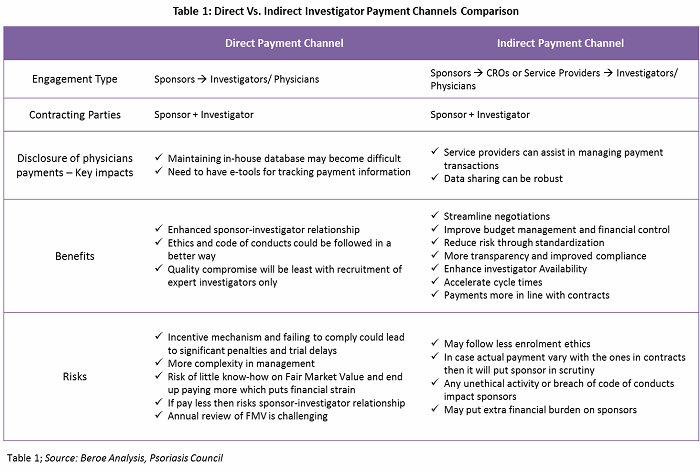
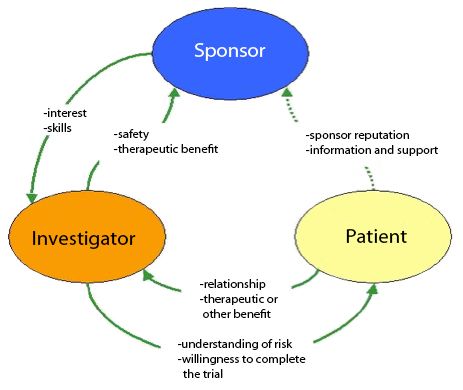
Frontiers | The Current Status and Future Direction of Clinical Research in Japan From a Regulatory Perspective
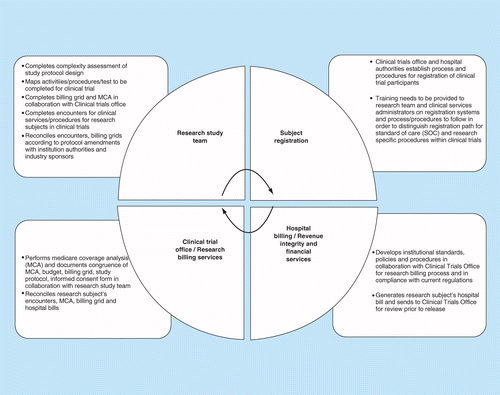
Optimization of protocol design: a path to efficient, lower cost clinical trial execution | Future Science OA

EORTC EU Clinical Trials Directives Organisation and Implementation of Cancer Clinical Trials Anastassia Negrouk EORTC Regulatory Affairs Manager Intergroup. - ppt download
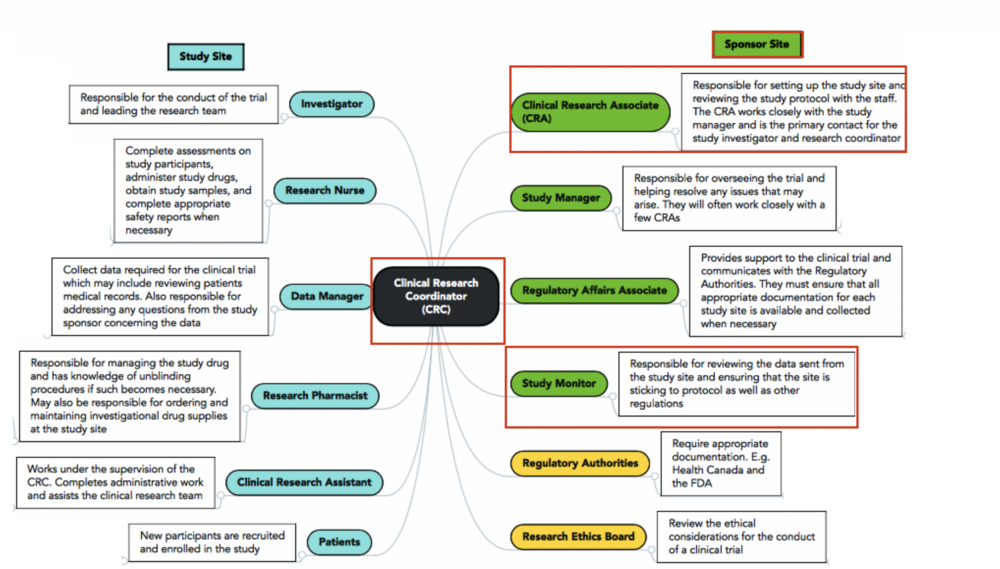
clinical research associate vs coordinator — Clinical Research Blog | Certified Clinical Research Professionals Society - Clinical Research Certification

WHEN, WHY AND HOW SPONSOR, CONTRACT RESEARCH ORGANISATION (CROs) AND RESEARCH SITES WORK TOGETHER: – your pharmacy guide