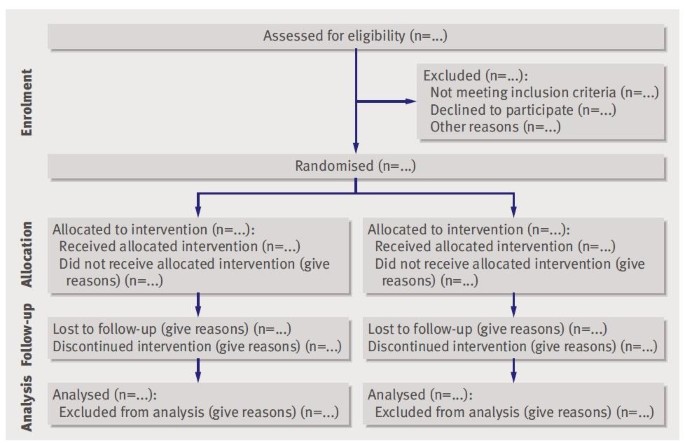
Poor patient-reported outcomes reporting according to CONSORT guidelines in randomized clinical trials evaluating systemic cancer therapy - Annals of Oncology

Figure 1 from Extending the CONSORT statement to randomized trials of nonpharmacologic treatment: explanation and elaboration. | Semantic Scholar

Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension - The Lancet Digital Health

Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension - ScienceDirect

Example of modified CONSORT flow diagram for individual randomized,... | Download Scientific Diagram

JCI - Randomized trial of calcipotriol combined with 5-fluorouracil for skin cancer precursor immunotherapy

CONSORT-SPI 2018 Explanation and Elaboration: guidance for reporting social and psychological intervention trials | Trials | Full Text

Compliance to the CONSORT Statement on Participant Flow Diagrams in Infectious Disease Randomized Clinical Trials | Semantic Scholar

Active implementation strategy of CONSORT adherence by a dental specialty journal improved randomized clinical trial reporting - ScienceDirect

Table 1 from Extending the CONSORT statement to randomized trials of nonpharmacologic treatment: explanation and elaboration. | Semantic Scholar
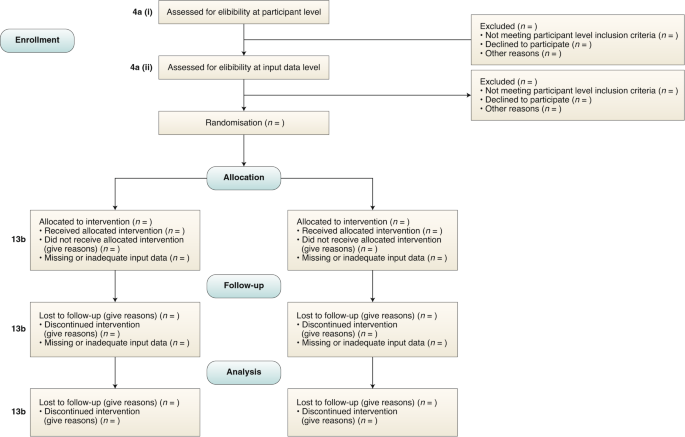
Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension | Nature Medicine

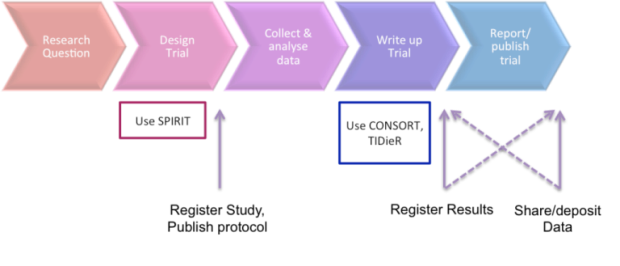
The CONSORT statement: revised recommendations for improving the quality of reports of parallel-group randomised trials - The Lancet
CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomised Trials | PLOS Medicine