EudraLex - Volume 4 - Good Manufacturing Practice (GMP) guidelines | BIOAIR S.p.a. - A Tecniplast Company | Advanced Therapy Isolator

The Handbook of Basic GMP Requirements: Collected guidelines from Eudralex Volume 4, Part I “Basic Requirements for Medicinal Products”: Santoro, Karyn Noemi: 9798552183685: Amazon.com: Books
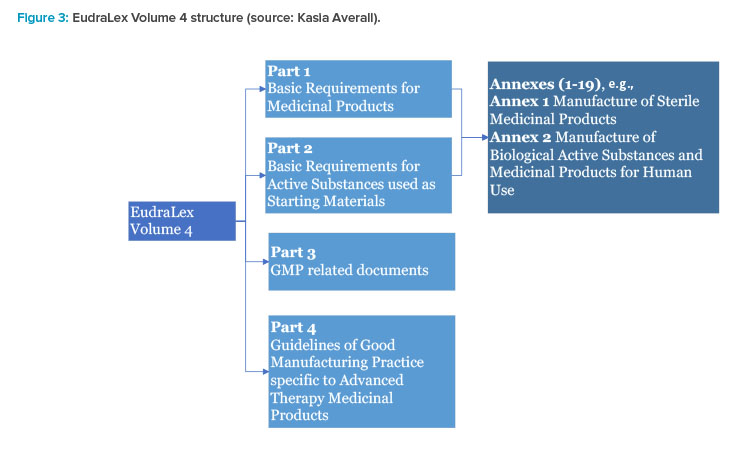
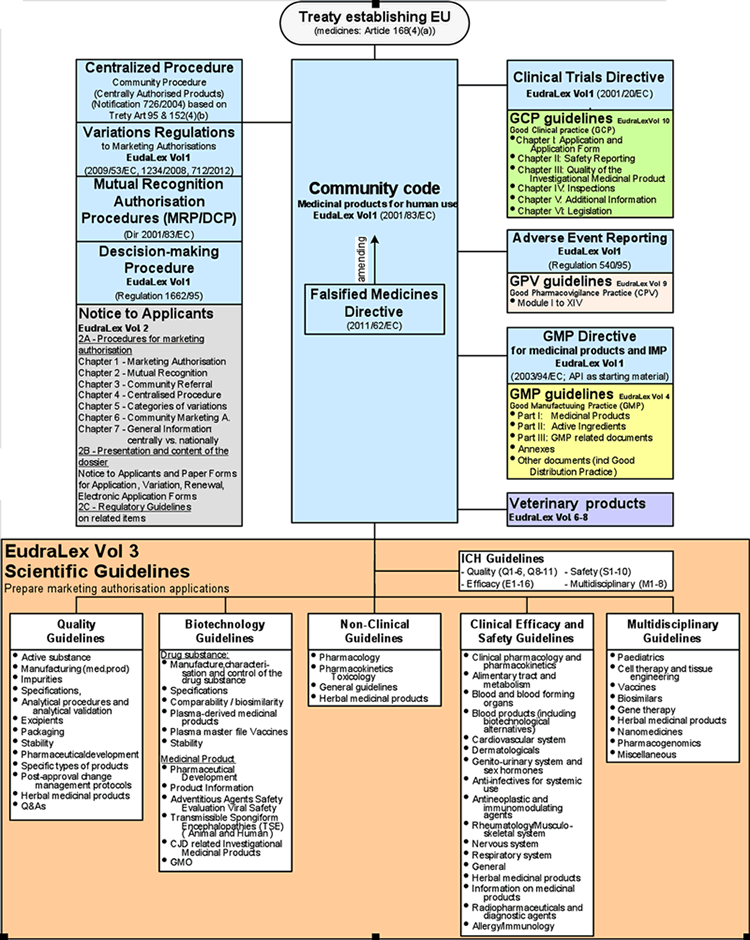
EUROPEAN COMMISSION EudraLex The Rules Governing Medicinal Products in the European Union Volume 4 EU Guidelines for Good Manuf

PDF) Good Manufacturing Practice (GMP) Guidelines (Eudralex-Volume 4) of the Creative Commons Attribution License (CC BY 4.0)

PPT - REVISION OF EUDRALEX VOL. 4 - GMP Luisa Stoppa, Ph.D. Inspection and Certification Department PowerPoint Presentation - ID:6720277
EUROPEAN COMMISSION Brussels, 13 August 2014 Ares(2014)2674284 EudraLex The Rules Governing Medicinal Products in the European U

GMP for medicinal products for human and veterinary use laid down in Commission Directives 91/356/EEC | M A N O X B L O G

Good Manufacturing Practice (GMP) Guidelines: The Rules Governing Medicinal Products in the European Union, EudraLex Volume 4 Concise Reference: Allport-Settle, Mindy J.: 9780982147603: Amazon.com: Books

EMA EudraLex - Volume 4 - GMP Guidelines - TELUGU GMP - Provides GMP Pharmaceutical Guidelines in Telugu.