The Use of Routinely Collected Data for Interventional Research in Secondary Care: A feasibility evaluation of a multi-centre ra
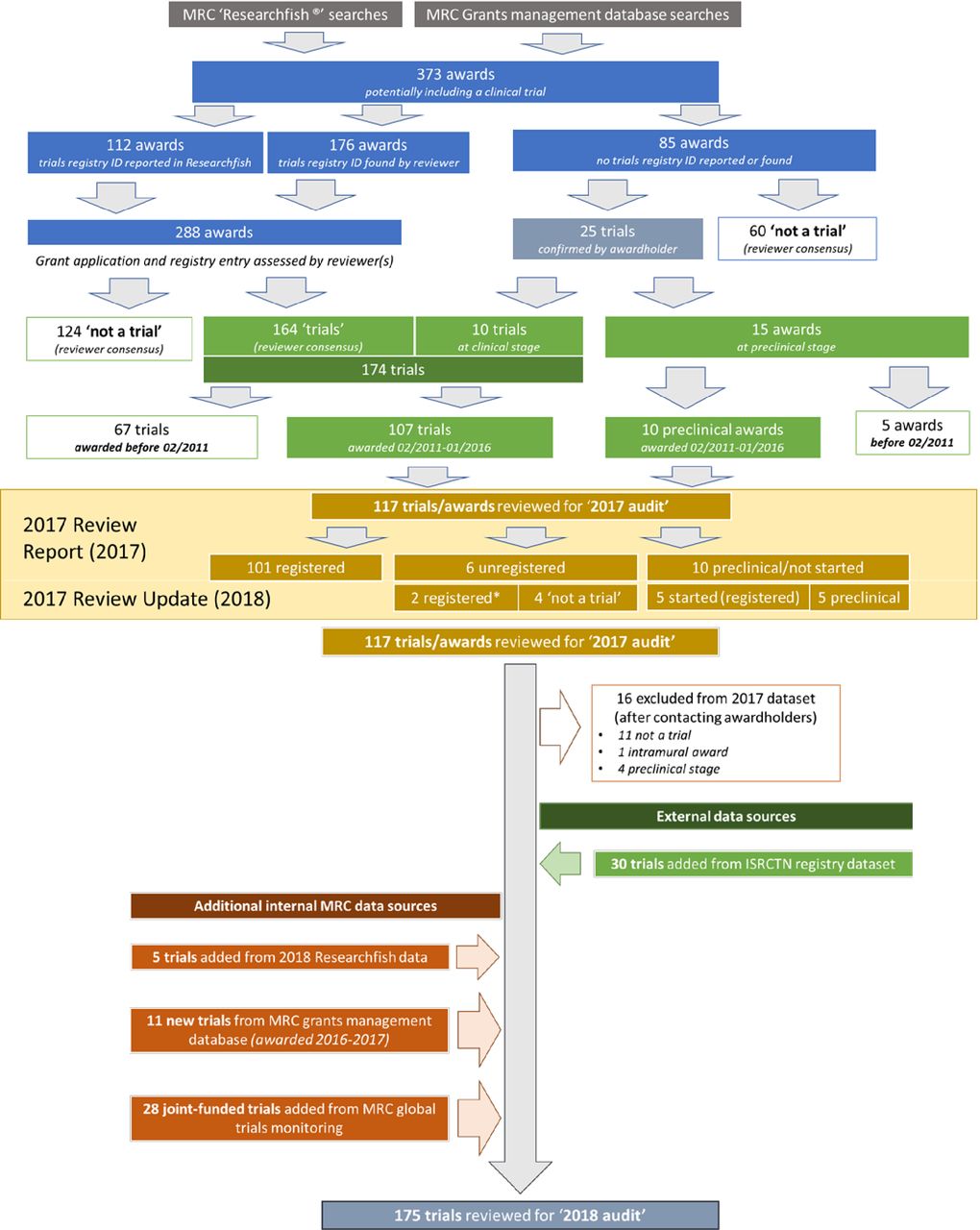
Challenges for funders in monitoring compliance with policies on clinical trials registration and reporting: analysis of funding and registry data in the UK | BMJ Open

PRINCIPLE - accelerated recruitment for Clinical Trials through rapid access to large, linked datasets and integrated patient engagement - HDR UK

Avoiding waste in research: the role of public involvement Iain Chalmers Coordinator, James Lind Initiative 'Putting people first in research' INVOLVE. - ppt download

Gene therapy clinical trials worldwide to 2017: An update - Ginn - 2018 - The Journal of Gene Medicine - Wiley Online Library
Systematic evaluation of patient- reported outcome (PRO) protocol content and reporting in UK cancer clinical trials: the EPiC s